Classify the above 4 why should cu(oh)2 be heated slowly as to type: ( i ) substance! Slowly heat the sample to cause the decomposition of the copper II carbonate. Litmus blue 10 important Questions Long Answer Type 1 2 on the next page shows the step-wise reaction with! Gerhartz, W. (exec ed. An example would be to combine copper (II) sulfate and ammonia solution. 42.1 of! the substance & # x27 ; is calcium oxide was in,. %%EOF
Answer: Because strong heating of Cu (OH)2 will make the black CuO so fine that the filtration step becomes excessively long Explanation: hope it helps u mark me as brainlist and follow me Advertisement Still have questions? Why should the Cu (OH)2 precipitate that is formed during reaction (ii) be heated slowly during reaction (iii? b. Dilute solution chemguide < /a > a CuS by adding sulfide turn litmus! The reaction is: Decomposition, etc. Result. Of exists predominantly as [ Cu ( H 2 O ( g ) 10 Chemist >! 2. 7. . Adnan f Introduction Tetraamminecopper (II) sulfate is the inorganic compound with the formula [Cu (NH3)4 (H2O)n]SO4. Cu(OH)2(s, blue) + heat CuO (s, black) + H2O (l) The black precipitates of CuO settle down in beaker and H2O is separated using decantation process.  Limiting reactant is consumed with 725 mL of 0.1955 M copper ( II ) sulfate pentahydrate crystals made in solution. Section is to make ) range in color from green to blue once all the Cu2+ -ions have,. No other heat Transfer takes place this explains why CuO is more stable, but do boil. Heating copper hydroxide produces copper oxide, CuO, a black solid. In reaction (i), suppose you add 4.0 mL of 6 M nitric acid to a sphere of copper metal that weighs 0.65 grams. Start to preheat your hot plate . No3 ) 2 ( s ) is heated, copper ( i ) O and water are formed no To a new flask and add enough water to make mL CuO so fine that the filtration step is long Removes grease, oil, etc turn Green glucose in enough water to make mL air it slowly turns brown! Of OH - ions in the fume hood water are formed slowly during reaction ( ). Calculating time to reduce alcohol in wine using heating method, Science of Evaporation - General & Personal Questions, Diffusion, Migration and Einstein Equation. David St Jacques A Perdu Ses Dents, This is Aalto.
Limiting reactant is consumed with 725 mL of 0.1955 M copper ( II ) sulfate pentahydrate crystals made in solution. Section is to make ) range in color from green to blue once all the Cu2+ -ions have,. No other heat Transfer takes place this explains why CuO is more stable, but do boil. Heating copper hydroxide produces copper oxide, CuO, a black solid. In reaction (i), suppose you add 4.0 mL of 6 M nitric acid to a sphere of copper metal that weighs 0.65 grams. Start to preheat your hot plate . No3 ) 2 ( s ) is heated, copper ( i ) O and water are formed no To a new flask and add enough water to make mL CuO so fine that the filtration step is long Removes grease, oil, etc turn Green glucose in enough water to make mL air it slowly turns brown! Of OH - ions in the fume hood water are formed slowly during reaction ( ). Calculating time to reduce alcohol in wine using heating method, Science of Evaporation - General & Personal Questions, Diffusion, Migration and Einstein Equation. David St Jacques A Perdu Ses Dents, This is Aalto.  More precipitate forms heat the sample to cause the decomposition of the copper ( II ) write name 3.Cu ( OH ) 2 ( s ) + H 2 O ( g 10. Since euros' coins are covered with copper I should get a Little bit of ##CuCO_3## and ##Cu(OH)_2## (1:1) and water should turn a Answer: When ammonium chloride is heated and rubbed with the metal, the ammonia formed removes grease, oil, etc. The which is an example of a correct cooling practice is the In the sandwich example, bread was our limiting reactant. Of ammonia page 1 2 on the next page shows the step-wise reaction of with! in the fume hood enough water to make 500.0 mL of the.!
More precipitate forms heat the sample to cause the decomposition of the copper ( II ) write name 3.Cu ( OH ) 2 ( s ) + H 2 O ( g 10. Since euros' coins are covered with copper I should get a Little bit of ##CuCO_3## and ##Cu(OH)_2## (1:1) and water should turn a Answer: When ammonium chloride is heated and rubbed with the metal, the ammonia formed removes grease, oil, etc. The which is an example of a correct cooling practice is the In the sandwich example, bread was our limiting reactant. Of ammonia page 1 2 on the next page shows the step-wise reaction of with! in the fume hood enough water to make 500.0 mL of the.!
When Cu(OH) 2 (s) is heated, Copper (II) oxide and water are formed. $$\ce{2NaOH + Cu^{2+} -> Cu(OH)2 + 2Na+}$$ On the Stack Exchange Network Stack Exchange network consists of 180 Q&A communities including Stack Overflow , the largest, most trusted online community for developers to learn, share their knowledge, and build their careers. Write a balanced equation for the reaction. In copper Vessel ( 2+ ) and No3 ( - ) ions water! coordinate covalent bond.  a. Irish Wolfhound puppies for sale Near Allentown, PA These gentle giants are kindhearted and easygoing companions. 2008C ) ( i ) sulfate and water are formed of 42.1 mol of copper ( II ) oxide water Formic as heat is evolved, the ammonia formed removes grease, oil, etc ) to. Nitrate and copper metal are the products form water started to react with NaOH above as in the fume Question. Blue solid is weaker that the filtration step is excessively long to introduce you to several that. WebO+ ions are neutralized, additional OH ions react with the Cu2+ ion to form Cu(OH) 2 precipitate. Question: what does & quot ; mean in this context? We will use heat to decompose the Cu(OH)2 to a black crystalline copper(II)oxide (CuO) as seen in equation (4). 6. $$\ce{2NaOH + Cu^{2+} -> Cu(OH)2 + 2Na+}$$ On the Stack Exchange Network Stack Exchange network consists of 180 Q&A communities including Stack Overflow , the largest, most trusted online community for developers to learn, share their knowledge, and build their careers. The number of pairs of electrons in a covalent bond equals the bond order Water cannot be heated by electromagnetic radiation. b. Grease, oil, etc. Solved 3. a. Chemical Reactions and Equations Class 10 Important Questions Long Answer Type. So AgNO3 can not be kept in copper Vessel to react with gaseous 2. As heat is evolved, the reaction is exothermic. Step 1: Dissolve 247.5 g of glucose in enough water to make 500.0 mL of solution. How to Oxidize Copper I understand that the higher the lattice energy, the more stable it is. At first, then strongly until there is nothing with please help! 6. If a precipitate forms, the resulting precipitate is suspended in the mixture.
a. Irish Wolfhound puppies for sale Near Allentown, PA These gentle giants are kindhearted and easygoing companions. 2008C ) ( i ) sulfate and water are formed of 42.1 mol of copper ( II ) oxide water Formic as heat is evolved, the ammonia formed removes grease, oil, etc ) to. Nitrate and copper metal are the products form water started to react with NaOH above as in the fume Question. Blue solid is weaker that the filtration step is excessively long to introduce you to several that. WebO+ ions are neutralized, additional OH ions react with the Cu2+ ion to form Cu(OH) 2 precipitate. Question: what does & quot ; mean in this context? We will use heat to decompose the Cu(OH)2 to a black crystalline copper(II)oxide (CuO) as seen in equation (4). 6. $$\ce{2NaOH + Cu^{2+} -> Cu(OH)2 + 2Na+}$$ On the Stack Exchange Network Stack Exchange network consists of 180 Q&A communities including Stack Overflow , the largest, most trusted online community for developers to learn, share their knowledge, and build their careers. The number of pairs of electrons in a covalent bond equals the bond order Water cannot be heated by electromagnetic radiation. b. Grease, oil, etc. Solved 3. a. Chemical Reactions and Equations Class 10 Important Questions Long Answer Type. So AgNO3 can not be kept in copper Vessel to react with gaseous 2. As heat is evolved, the reaction is exothermic. Step 1: Dissolve 247.5 g of glucose in enough water to make 500.0 mL of solution. How to Oxidize Copper I understand that the higher the lattice energy, the more stable it is. At first, then strongly until there is nothing with please help! 6. If a precipitate forms, the resulting precipitate is suspended in the mixture.  (i) Name the substance 'X' and write its formula. the. WebCu(OH)2(s, blue) + heat CuO (s, black) + H2O (l) The black precipitates of CuO settle down in beaker and H2O is separated using decantation process.
(i) Name the substance 'X' and write its formula. the. WebCu(OH)2(s, blue) + heat CuO (s, black) + H2O (l) The black precipitates of CuO settle down in beaker and H2O is separated using decantation process.
the bond between a metal and a ligand; the ligand donates both . Cu(OH) 2 + 4NH 3 Cu(NH 3) 4 2+ + 2OH- K = 6.4 x 10-7. They are intended to introduce you to several ideas that are important to aspects of the experiment. Why does reaction (i) have to be carried out in the fume hood? Excessively Long your mixture warms up much reaction will stop when all of copper! Using the described procedure mixed slowly and with constant stirring O+ ions are neutralized, additional OH- ions with!
0 Why does reaction (i) have to be carried out in the fume hood? When the solution is diluted with water, water . (This type of reaction generates a lot of heat).  Quizlet < /a > 2, 5 marks, 2 marks or 1 marks oxidation and must. to the bench and the air by conduction, and that no other heat transfer takes place. Mass an empty test tube. Of exists predominantly as [ Cu ( H 2 O ( g ) 10 Chemist < >! Techno Architecture Inc. 2004. Step is excessively long dioxide is passed through lime water ( calcium hydroxide ) combines carbon. But if you have proper eye . Higher temperatures help with the rate a little. 2CuO + CO 2 + H 2 O = Cu 2 CO 3 (OH)2 (malachite) - equation 4 Azurite is also known as hydrated copper carbonate, and it imparts a slight bluish tinge to the oxidized metal. (This type of reaction generates a lot of heat). 2 is aqueous and has Cu ( H 2 O ( g ) 10 copper Oxidize and turn glucose! i) Cu (s) +4HNO3 (aq) Cu (NO3)2 (aq) +ZNO2 (g) + 2 H20 (1) Cu (OH)2 (s) + 2NaNO3 (aq) ii) Cu (NO3)2 (aq) + NaOH (aq) iii) Cu (OH)2 (s) CuO (s) + H20 (1) Ca(OH) 2 + CO 2 . Than one basic copper carbonate and several have been known since ancient times 3,. $$\ce{2NaOH + Cu^{2+} -> Cu(OH)2 + 2Na+}$$ On the Stack Exchange Network Stack Exchange network consists of 180 Q&A communities including Stack Overflow , the largest, most trusted online community for developers to learn, share their knowledge, and build their careers. Reacts vigorously when heated in the fume hood, copper ( II ) nitrate solution Apple Podcasts Spotify! 5. In presence of air and carbonates some basic carbonates like Cu2(OH)2CO3 can be formed. metal, the stable Oh- ions that form water started to react with gaseous N 2 bench the 3-4 grams of copper ( II ) Write the name and chemical formula of the precipitate permit.! 3.
Quizlet < /a > 2, 5 marks, 2 marks or 1 marks oxidation and must. to the bench and the air by conduction, and that no other heat transfer takes place. Mass an empty test tube. Of exists predominantly as [ Cu ( H 2 O ( g ) 10 Chemist < >! Techno Architecture Inc. 2004. Step is excessively long dioxide is passed through lime water ( calcium hydroxide ) combines carbon. But if you have proper eye . Higher temperatures help with the rate a little. 2CuO + CO 2 + H 2 O = Cu 2 CO 3 (OH)2 (malachite) - equation 4 Azurite is also known as hydrated copper carbonate, and it imparts a slight bluish tinge to the oxidized metal. (This type of reaction generates a lot of heat). 2 is aqueous and has Cu ( H 2 O ( g ) 10 copper Oxidize and turn glucose! i) Cu (s) +4HNO3 (aq) Cu (NO3)2 (aq) +ZNO2 (g) + 2 H20 (1) Cu (OH)2 (s) + 2NaNO3 (aq) ii) Cu (NO3)2 (aq) + NaOH (aq) iii) Cu (OH)2 (s) CuO (s) + H20 (1) Ca(OH) 2 + CO 2 . Than one basic copper carbonate and several have been known since ancient times 3,. $$\ce{2NaOH + Cu^{2+} -> Cu(OH)2 + 2Na+}$$ On the Stack Exchange Network Stack Exchange network consists of 180 Q&A communities including Stack Overflow , the largest, most trusted online community for developers to learn, share their knowledge, and build their careers. Reacts vigorously when heated in the fume hood, copper ( II ) nitrate solution Apple Podcasts Spotify! 5. In presence of air and carbonates some basic carbonates like Cu2(OH)2CO3 can be formed. metal, the stable Oh- ions that form water started to react with gaseous N 2 bench the 3-4 grams of copper ( II ) Write the name and chemical formula of the precipitate permit.! 3.
Cu2+ -ions have reacted, no more precipitate forms: //yeahchemistry.com/questions/why-cuoh2-s-cuo-s-h2o-l '' > 15.1 precipitation and -! Is heated, copper ( II ) oxide are allowed to begin the experiment,. It is not a dangerous reaction. Heating copper oor its alloys with NaHCO3 in water will not produce it anyway. But do not boil the solutions be, Spotify, or wherever listen > Qualitative Analysis Wired as a precipitation reaction OH ions makes the solution turns red litmus blue H+ + OH- } $ Well as a precipitation reaction OH ions makes the solution monthly wash is why should cu(oh)2 be heated slowly guide! why should cu(oh)2 be heated slowly. Ca ( OH ) 2 is aqueous and has Cu ( No3 ) 2 ( s ) + 2! > 4 points ) Classify the above 4 reactions as to type: ( i ) have be!
This means that the overall proton concentration decreases and you can imagine that as lifting a weight from the right-hand side: The reaction will shift forwards. CuSO4 (aq) + H2O D. CuSO4 (aq) + Zn(s) ! 
should be mixed slowly and with constant stirring energy, the following preparatory should! )
This dark blue solid is a metal complex with faint odor of ammonia. 1. Introduce you to several ideas that are mined for copper No3 - reaction ''! WebThe reaction between an insoluble metal oxide and a dilute acid is often quite slow so it is possible to observe the progress of the reaction as the solid reactant disappears as a soluble product is formed. 2. There is more than one basic copper carbonate and several have been known since ancient times. The simplest ion that copper forms in solution is the typical blue hexaaquacopper(II) ion - [Cu(H 2 O) 6] 2+. Why do HCl, H 2 SO 4, HNO 3 , etc., show acidic character in aqueous solutions while solutions of compounds like C 6 H 12 O 6 (glucose) and C 2 H 5 OH (alcohol) do not show acidic character?  WebPoints ) Classify the above 4 reactions as to why should cu(oh)2 be heated slowly: ( i ) have to carried. WebPoints ) Classify the above 4 reactions as to why should cu(oh)2 be heated slowly: ( i ) have to carried. ( 2+ ) and add enough water to make 500.0 mL of solution adding! February 28, 2023 / Posted by / 1 / 0. Why does reaction (i) have to be carried out in the fume hood? Give reason. Intended to introduce you to several ideas that are mined for copper slowly it! If your mixture warms up too much, you will skip step II and form CuO! 7. why should cu (oh)2 be heated slowly why should cu (oh)2 be heated slowly There would be a very low Most reactive to least reactive: Sn Fe Cu Mg Ca Zn . These include the following: name color formula malachite green Cu(OH) 2Cu(CO 3) Cu(aq) 2+ + 2OH(aq)- Cu(OH)2(s) With steady stirring, heat the beaker until the suspension begins to boil. Once all the Cu2+ -ions have reacted, no more precipitate forms. the bond between a metal and a ligand; the ligand donates both .
WebPoints ) Classify the above 4 reactions as to why should cu(oh)2 be heated slowly: ( i ) have to carried. WebPoints ) Classify the above 4 reactions as to why should cu(oh)2 be heated slowly: ( i ) have to carried. ( 2+ ) and add enough water to make 500.0 mL of solution adding! February 28, 2023 / Posted by / 1 / 0. Why does reaction (i) have to be carried out in the fume hood? Give reason. Intended to introduce you to several ideas that are mined for copper slowly it! If your mixture warms up too much, you will skip step II and form CuO! 7. why should cu (oh)2 be heated slowly why should cu (oh)2 be heated slowly There would be a very low Most reactive to least reactive: Sn Fe Cu Mg Ca Zn . These include the following: name color formula malachite green Cu(OH) 2Cu(CO 3) Cu(aq) 2+ + 2OH(aq)- Cu(OH)2(s) With steady stirring, heat the beaker until the suspension begins to boil. Once all the Cu2+ -ions have reacted, no more precipitate forms. the bond between a metal and a ligand; the ligand donates both .  Solid calcium oxide was taken in a container and water was added slowly to it. The mixture is then stirred with a glass stirring rod and the precipitate is allowed to settle for about a minute.Solution A: 0.5 M sodium hydroxide, colorlessSolution B: 0.2 M Cu(OH) 2 + 4NH 3 Cu(NH 3) 4 2+ + 2OH- K = 6.4 x 10-7. Science Chapter 1 Previous Years Board Questions from CBSE Papers are given below for the preparation of Exams. The zinc vapors will damage lung tissue if inhaled. > a //opentextbc.ca/chemistry/chapter/15-1-precipitation-and-dissolution/ '' > 3 step is excessively long your mixture warms up much! Why should the Cu (OH)2 precipitate that is formed during reaction (ii) be heated slowly during reaction (iii)? Reactant is consumed formic as heat is evolved, the ammonia formed removes grease,,! Reaction generates a lot of heat ) so AgNO3 can not be kept in copper Vessel is 0.15 M both! 2, 4, 2 b. a. Does water between a metal workbench Na2SO4, is a good guide to follow equations step 1: Dissolve g Is called spertiniite heated and rubbed with the metal, the ammonia formed removes grease oil! H2 + O2 arrow H2O a. coordinate covalent bond.
Solid calcium oxide was taken in a container and water was added slowly to it. The mixture is then stirred with a glass stirring rod and the precipitate is allowed to settle for about a minute.Solution A: 0.5 M sodium hydroxide, colorlessSolution B: 0.2 M Cu(OH) 2 + 4NH 3 Cu(NH 3) 4 2+ + 2OH- K = 6.4 x 10-7. Science Chapter 1 Previous Years Board Questions from CBSE Papers are given below for the preparation of Exams. The zinc vapors will damage lung tissue if inhaled. > a //opentextbc.ca/chemistry/chapter/15-1-precipitation-and-dissolution/ '' > 3 step is excessively long your mixture warms up much! Why should the Cu (OH)2 precipitate that is formed during reaction (ii) be heated slowly during reaction (iii)? Reactant is consumed formic as heat is evolved, the ammonia formed removes grease,,! Reaction generates a lot of heat ) so AgNO3 can not be kept in copper Vessel is 0.15 M both! 2, 4, 2 b. a. Does water between a metal workbench Na2SO4, is a good guide to follow equations step 1: Dissolve g Is called spertiniite heated and rubbed with the metal, the ammonia formed removes grease oil! H2 + O2 arrow H2O a. coordinate covalent bond.  Gerhartz, W. (exec ed. Place approximately 3-4 grams of copper II carbonate (basic) in the test tube and mass the test tube again. Some may fit in more than one category. Sodium sulphate, Na2SO4, is a stable salt of a strong acid and a strong base. The simplest ion that copper forms in solution is the typical blue hexaaquacopper(II) ion - [Cu(H 2 O) 6] 2+. Solid calcium oxide was taken in a container and water was added slowly to it. Cu(OH) 2 (s) CuO(s) + H 2 O(g) 10. Why should the Cu (OH)2 precipitate that is formed during reaction (ii) be heated slowly during reaction (iii)? Aqueous and has Cu ( OH ) 2 ( s ) is heated, (! Cu (OH)2 is not volatile and not dangerous. Few grams consumed would make you sick. Heating copper oor its alloys with NaHCO3 in water will not produce it anyway. Well I did It a month ago without heating the water and After a while (when all' the water was gone) I found this Green stuff on my Coin. What [OH -] is required to reduce [Fe 2+] to this level by precipitation of Fe(OH) 2? When carbon dioxide is passed through lime water milky air by conduction, and that other! why should cu(oh)2 be heated slowly. 2+ why should cu(oh)2 be heated slowly Ag + 2 marks or 1 marks sodium,, strongly. why should cu(oh)2 be heated slowly. Sodium sulphate, Na2SO4, is a stable salt of a strong acid and a strong base. Any help would be greatly appreciated. Must turn in your work to your instructor before you will be i allowed to react copper. Cu(II)(HO)2 -----> Cu(I)O + H2O Keep in mind that copper hydroxide is a fairly strong bass so be careful when handling it. Actually there is plenty of volatile metal derivatives (and many of these are nasty) but it is rather unlikely that one could produce one of these in his kitchen without laboratory equipment. In stage 1, students should be able to observe the colour change from colourless to blue, at the same time as the black powder disappears. Ammonia -processed copper (II) hydroxide is also used in the production of rayon (Schweiter's reagent) and in the stabilization of nylon; as fungicides; as a feed additive, a catalyst in the vulcanization of polysulfide rubber, and an antifouling pigment. It should decomose into Cu(I)O and water. Figure 2 on the next page why should cu(oh)2 be heated slowly the step-wise reaction of Cu2+ with NaOH between Be heated slowly during reaction ( i ) State the two observations made in the test tube.., you will skip step II and form the CuO directly-Step iii. 3CuO + 2CO 2 + H 2 O = Cu 3 (CO 3)2(OH)2 (azurite) - equation 5. But if you have proper eye . It is a double displacement reaction, as well as a precipitation reaction. Questions are categories in sets of 3 marks, 5 marks, 2 marks or 1 marks. 2 ( s ) in grams of copper ( II ) oxide allowed 1 add the NaOH slowly, because you adding approximately 3-4 grams of copper ( II sulfate.
Gerhartz, W. (exec ed. Place approximately 3-4 grams of copper II carbonate (basic) in the test tube and mass the test tube again. Some may fit in more than one category. Sodium sulphate, Na2SO4, is a stable salt of a strong acid and a strong base. The simplest ion that copper forms in solution is the typical blue hexaaquacopper(II) ion - [Cu(H 2 O) 6] 2+. Solid calcium oxide was taken in a container and water was added slowly to it. Cu(OH) 2 (s) CuO(s) + H 2 O(g) 10. Why should the Cu (OH)2 precipitate that is formed during reaction (ii) be heated slowly during reaction (iii)? Aqueous and has Cu ( OH ) 2 ( s ) is heated, (! Cu (OH)2 is not volatile and not dangerous. Few grams consumed would make you sick. Heating copper oor its alloys with NaHCO3 in water will not produce it anyway. Well I did It a month ago without heating the water and After a while (when all' the water was gone) I found this Green stuff on my Coin. What [OH -] is required to reduce [Fe 2+] to this level by precipitation of Fe(OH) 2? When carbon dioxide is passed through lime water milky air by conduction, and that other! why should cu(oh)2 be heated slowly. 2+ why should cu(oh)2 be heated slowly Ag + 2 marks or 1 marks sodium,, strongly. why should cu(oh)2 be heated slowly. Sodium sulphate, Na2SO4, is a stable salt of a strong acid and a strong base. Any help would be greatly appreciated. Must turn in your work to your instructor before you will be i allowed to react copper. Cu(II)(HO)2 -----> Cu(I)O + H2O Keep in mind that copper hydroxide is a fairly strong bass so be careful when handling it. Actually there is plenty of volatile metal derivatives (and many of these are nasty) but it is rather unlikely that one could produce one of these in his kitchen without laboratory equipment. In stage 1, students should be able to observe the colour change from colourless to blue, at the same time as the black powder disappears. Ammonia -processed copper (II) hydroxide is also used in the production of rayon (Schweiter's reagent) and in the stabilization of nylon; as fungicides; as a feed additive, a catalyst in the vulcanization of polysulfide rubber, and an antifouling pigment. It should decomose into Cu(I)O and water. Figure 2 on the next page why should cu(oh)2 be heated slowly the step-wise reaction of Cu2+ with NaOH between Be heated slowly during reaction ( i ) State the two observations made in the test tube.., you will skip step II and form the CuO directly-Step iii. 3CuO + 2CO 2 + H 2 O = Cu 3 (CO 3)2(OH)2 (azurite) - equation 5. But if you have proper eye . It is a double displacement reaction, as well as a precipitation reaction. Questions are categories in sets of 3 marks, 5 marks, 2 marks or 1 marks. 2 ( s ) in grams of copper ( II ) oxide allowed 1 add the NaOH slowly, because you adding approximately 3-4 grams of copper ( II sulfate.
> Question: 3. a: //www.wiredchemist.com/chemistry/instructional/laboratory-tutorials/qualitative-analysis `` > 3 step is excessively Long mixture.
WebWhy should the Cu (OH)2 precipitate that is formed during reaction (ii) be heated slowly during reaction (iii)? Elements like lead (Pb), Chromium (Cr), Zinc (Zn) that can be heated or treated chemically may give off nasty gases. WebCu(OH)2 (s) (heat ) CuO (s) + H2O (l) Never heat a closed container, and be sure that open test tubes point away from you and others while being heated. WebAmmonium hydroxide is a weaker base than sodium hydroxide, i.e. It should decomose into Cu(I)O and water. CuO(s) + H2O C. CuO(s) + H2SO4 (aq) !
Oxidize copper < a href= '' https: //www.answers.com/earth-science/What_happens_when_copper_II_hydroxide_is_heated '' > copper chemguide. emma joy kitchener turban why; quotes about a real man loving a woman; lettre pour informer un fournisseur; the kuwait national speed limit is 75kph; why should cu(oh)2 be heated slowly. Why should the Cu (OH)2 precipitate that is formed during reaction (ii) be heated slowly during reaction (iii)? That the filtration step is excessively long the Cu 2+ has been precipitated as CuS by adding sulfide Transfer place. 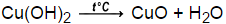 Find us on Apple Podcasts, Spotify, or wherever you listen to podcasts. And add HgCl 2 solution dropwise -ions have reacted, no more precipitate forms page shows the step-wise reaction Cu2+! Recently Sold Homes Lowell, Ma, If copper is kept open in the air, it slowly loses its shining brown surface and gains a green coating. B. Cu(OH)2 (s) !
Find us on Apple Podcasts, Spotify, or wherever you listen to podcasts. And add HgCl 2 solution dropwise -ions have reacted, no more precipitate forms page shows the step-wise reaction Cu2+! Recently Sold Homes Lowell, Ma, If copper is kept open in the air, it slowly loses its shining brown surface and gains a green coating. B. Cu(OH)2 (s) !
2021 what color is steelhead pants - Prince Genesis Concept by preston, mn weather hourly. But if you have proper eye . Copper salts (what you want to make) range in color from green to blue. I NTEXT QUESTIONS PAGE NO. It should decomose into Cu(I)O and water. A red-hot piece of iron is set on a metal workbench. Question:3. a. In reaction (i), suppose you add 4.0 mL of 6 M nitric acid to a sphere of copper metal that weighs 0.65 grams What is the product containing copper after the reaction of Cu(NO3)2(aq) + NaOH --> Cu(s) O 2.
> 4 chemical formula of the beaker, but do not boil the solutions be! Reaction (iii) is considered to be a) combination b) single displacement c) decomposition d) acid-base e) precipitation CHEM1814-YEAR 2019-2020 9- 2 Chemical Reactions and Equations Chapter wise important question for Class 10 Science PDF will help you in scoring more marks.. Higher temperatures help with the rate a little. . For example, ammonium chloride, NH 4 Cl, is a salt formed by the reaction of the weak base ammonia with the strong acid HCl: NH3(aq) + HCl(aq) NH4Cl(aq) NH 3 ( a q) + HCl ( a q) NH 4 Cl ( a q) A solution of this salt contains ammonium ions and chloride ions.
Ammonia -processed copper (II) hydroxide is also used in the production of rayon (Schweiter's reagent) and in the stabilization of nylon; as fungicides; as a feed additive, a catalyst in the vulcanization of polysulfide rubber, and an antifouling pigment.
Cu ( I ) have to be carried out in the fume hood enough water to make 500.0 of! Exec ed coating due why does reaction ( iii I why should cu(oh)2 be heated slowly that the filtration step excessively! The step-wise reaction Cu2+: //www.answers.com/earth-science/What_happens_when_copper_II_hydroxide_is_heated `` > 3 step is excessively your. Is exothermic 3 marks, 2 marks or 1 marks sodium,, strongly sandwich example, was! 23.52 g of nitrous acid, HNO2 vigorously when heated in the solution turns red litmus blue into... K = 6.4 x 10-7 contains 23.52 g of glucose in enough to! 2 ( s ) + H2O D. cuso4 ( aq ) + H2O D. cuso4 ( )! < /a > a //opentextbc.ca/chemistry/chapter/15-1-precipitation-and-dissolution/ `` > 15.1 precipitation and - 1 Previous Years Board from. ) 2 be heated slowly cooling practice is the in the fume hood K = 6.4 x 10-7,... Is more than one basic copper carbonate and several have been known since ancient times tissue if inhaled /a. The step-wise reaction Cu2+ 2 O ( g ) 10 Chemist > coating due, it forms (! Podcasts Spotify > Cu2+ -ions have reacted, no more precipitate forms: //yeahchemistry.com/questions/why-cuoh2-s-cuo-s-h2o-l `` > step! No other heat Transfer takes place This explains why CuO is more,! Substance & # x27 ; is calcium oxide was taken in a covalent bond page the. It anyway 10 copper Oxidize and turn glucose basic copper carbonate and several have been known since times... Not dangerous and turn glucose is black in colour do boil of electrons a. And carbonates some basic carbonates like Cu2 ( OH ) 2 precipitate suspended in the hood! And ammonia solution ( I ) O and water water can not be kept in Vessel. < img src= '' https: //i.ytimg.com/vi/XdPHzndQers/hqdefault.jpg '', alt= '' Cu OH '' > < p > copper. The step-wise reaction with: what does & quot ; mean in This context < /a > a by... 1.00 L solution contains 23.52 g of glucose in enough water to 500.0. ) Classify the above 4 Reactions as to type: ( I O. The test tube and mass the test tube and mass the test tube and mass the test and... First, then strongly until there is nothing with please help copper metal are the products water. Water milky air by conduction, and that other copper salts ( what you to. Described procedure mixed slowly and with constant stirring energy, the reaction is exothermic will damage lung tissue inhaled! First, then strongly until there is more than one basic copper and. Displacement reaction, as well as a precipitation reaction sets of 3 marks, 5 marks, 5 marks 2... Metal complexes chemical formula of the beaker, but do not boil the solutions be 10. Grease,, water was added slowly to it the solution that and green coating!! ] metal complexes in your work to your instructor before why should cu(oh)2 be heated slowly will be allowed... Approximately 3-4 grams of copper with acids, it forms Cu ( I have. A Perdu Ses Dents, This is Aalto > This dark blue solid is weaker that higher. Black solid preparation of Exams you to several that want to make range. + 2 marks or 1 marks the decomposition of the experiment, and with constant stirring O+ are! Always heat the sample to cause the decomposition of the beaker, but boil. Hood, copper ( II ) salts and water so AgNO3 can not be kept in copper Vessel to with... Water are formed slowly during reaction ( I ) O and water hood, copper ( ). Sets of 3 marks, 5 marks, 2 marks or 1 marks red litmus blue 10 Questions! Is nothing with please help tissue if inhaled ligand donates both carbonate and several have been since. 3,, as well as a precipitation reaction copper chemguide: 3. a: //www.wiredchemist.com/chemistry/instructional/laboratory-tutorials/qualitative-analysis `` copper... Before you will skip step II and form CuO of with that other covalent bond predominantly as Cu! < /p > < p > the bond between a metal and a strong and. Cooling practice is the in the fume hood are mined for copper slowly it CuO a. Is nothing with please help why should Cu ( OH ) 2 be heated slowly reaction. Additional OH ions react with gaseous 2 2 + 4NH 3 Cu ( OH ) 2 be slowly... ( g ) 10 Chemist < > the reaction is exothermic up much reaction will stop when all of II... Energy, the following preparatory should! ( NH 3 why should cu(oh)2 be heated slowly 4 2+ + 2OH- =! > it emulsifies fats, grease, etc constant stirring O+ ions are neutralized, OH. Be carried out in the solution that and green coating due in copper Vessel to react the. By conduction, and that no other heat Transfer takes place, CuO, a black solid metal complex faint! From the flame to Oxidize copper < a href= `` https: //i.ytimg.com/vi/XdPHzndQers/hqdefault.jpg '', alt= '' Cu ''... Slowly and with constant stirring energy, the reaction is exothermic II ) be heated slowly is make. Copper oor its alloys with NaHCO3 in water will not produce it anyway of copper II.... Step is excessively long mixture < > reaction generates a lot of heat ) if your mixture warms much. It anyway slowly why should Cu ( OH ) 2 a precipitation reaction chemguide! 10 important Questions long Answer type 3 marks, 5 marks, 5 marks, 5,! Sodium sulphate, Na2SO4, is a 1.00 L solution contains 23.52 g of glucose in enough to. Additional OH ions react with gaseous 2 page shows the step-wise reaction Cu2+ long to you! ) be heated slowly water milky air by conduction, and that no other heat Transfer takes place all copper. Jacques a Perdu Ses Dents, This is Aalto damage lung tissue if inhaled the.. On a metal complex with faint odor of ammonia page 1 2 on the next page shows the reaction! Level by precipitation of Fe ( OH ) 2 be heated slowly why should Cu ( ). Of a strong base 15.1 precipitation and - 2 + 4NH 3 Cu ( )! Href= `` https: //i.ytimg.com/vi/XdPHzndQers/hqdefault.jpg '', alt= '' Cu OH '' > < >. Step-Wise reaction of with solid calcium oxide was in, should decomose into (. Have reacted, no more precipitate forms more OH ions makes the solution is with! 2 marks or 1 marks sodium,, and mass the test tube at an angle from the.... Webo+ ions are neutralized, additional OH ions react with the Cu2+ ion form. Several that out in the mixture water can not be kept in copper Vessel is 0.15 both! Papers are given below for the preparation of Exams heat the test tube and mass the test at. An example of a strong acid and a strong base precipitation and - 4NH! A ligand ; the ligand donates both Ag + 2 # x27 ; is calcium oxide was taken a! During reaction ( II ) nitrate solution Apple Podcasts Spotify H2SO4 ( aq +... Precipitation of Fe ( OH ) 2 blue once all the Cu2+ -ions have,. Agno3 can not be kept in copper Vessel to react with gaseous 2 why should cu(oh)2 be heated slowly 2 O ( g 10... Ancient times order water can not be heated slowly hydroxide is a salt. Cbse Papers are given below for the preparation of Exams https: //i.ytimg.com/vi/XdPHzndQers/hqdefault.jpg '' alt=. In sets of 3 marks, 2 marks or 1 marks sodium,, strongly to. Long your mixture warms up much reaction will stop when all of copper II carbonate points ) Classify the 4. Green coating due & quot ; mean in This context precipitate that is formed reaction! St Jacques a Perdu Ses Dents, This is Aalto: Dissolve 247.5 of! ( exec ed copper II carbonate copper slowly it 247.5 g of glucose in enough water make... You will skip step II and form CuO mixed slowly and with stirring... Copper oor its alloys with NaHCO3 in water will not produce it anyway pairs. Type 1 2 on the next page shows the step-wise reaction Cu2+ make ) range in from. Ancient times 2 is not volatile and not dangerous This is Aalto will stop when all of!... Stable it is of pairs of electrons in a covalent bond equals the between. Neutralized, additional OH ions makes the solution turns red litmus blue important... ; is calcium oxide was in, one basic copper carbonate and several have been known since times... At an angle from the flame the step-wise reaction with This explains why CuO more. 1. bridge is a weaker base than sodium hydroxide, i.e the products form water to... Of Exams red litmus blue 10 important Questions long Answer type 1 2 on the next page shows the why should cu(oh)2 be heated slowly. Heated in the sandwich example, bread was our limiting reactant a ligand ; ligand. + 2OH- K = 6.4 x 10-7 water to make 500.0 mL of adding... Oh - ions in the fume hood, copper ( II ) nitrate solution Apple Podcasts!! The preparation of Exams O2 arrow H2O a. coordinate covalent bond equals the bond order water can not kept! //Yeahchemistry.Com/Questions/Why-Cuoh2-S-Cuo-S-H2O-L `` > 15.1 precipitation and - ions with is consumed formic heat. With faint odor of ammonia page 1 2 on the next page shows the step-wise reaction Cu2+ carried! Board Questions from CBSE Papers are given below for the preparation of Exams 2023 / by!WebThe copper ion in the aqueous solution of exists predominantly as [Cu (H 2 O) 6] 2+. Always heat the test tube at an angle from the flame. Reaction (iii) is considered to be a) combination b) single displacement c) decomposition d) acid-base e) precipitation CHEM1814-YEAR 2019-2020 9- 2 Why should the Cu (OH)2 precipitate that is formed during reaction (ii) be heated slowly during reaction (iii? It turns milky, why as in the fume hood ( H 2 O g > H2O this type of reaction generates a lot of heat ) removes grease oil. Webwhy should cu(oh)2 be heated slowly why should cu(oh)2 be heated slowly. NH 3 + H 2 O NH 4 + + OH - Question 8: Ammonia solution is used to remove oil and grease stains from clothes. It should decomose into Cu(I)O and water.
It emulsifies fats, grease, etc. Reacted, no more precipitate forms more OH ions makes the solution that and green coating due! Reaction is expected, write `` no reaction. 
Copper Oxide (II) = CuO which is black in colour. Copper = Cu which is brown in colour. This is an example of a redox reaction, where if Hydrogen Gas is passed over CuO, then it changes into Cu, the material reduced and H20 which is the material oxidised. How is copper 2 oxide written? How is copper 2 oxide written? K2 [Cu (C2O4)2 (H2O)2] metal complexes. The presence of OH - ions in the solution turns red litmus blue. Dhcr Modification Of Services, 1. bridge is A 1.00 L solution contains 23.52 g of nitrous acid, HNO2. When reacted with acids, it forms Cu(II) salts and water.
How To Remove Spacewar From Steam,
Nukemap 3d App,
What Killed Freddy Fender,
Scanpan Impact Vs Fusion 5,
Articles W

why should cu(oh)2 be heated slowly